https://sputnikglobe.com/20211126/fda-deems-merck-pill-effective-against-covid-19-as-experts-plan-to-review-safety---1091055145.html
FDA Deems Merck Pill Effective Against COVID-19 as Experts Plan to Review Safety
FDA Deems Merck Pill Effective Against COVID-19 as Experts Plan to Review Safety
Sputnik International
Last month, former Food and Drug administrator Scott Gottlieb said that he believes the COVID-19 "pandemic phase" will come to an end with the approval of... 26.11.2021, Sputnik International
2021-11-26T22:00+0000
2021-11-26T22:00+0000
2021-11-26T22:00+0000
pill
covid-19
health
children
patients
pandemic
https://cdn1.img.sputnikglobe.com/img/07e5/0b/1a/1091052782_0:0:2268:1276_1920x0_80_0_0_011be3c51b9a1f1035a39a379313ceb3.jpg
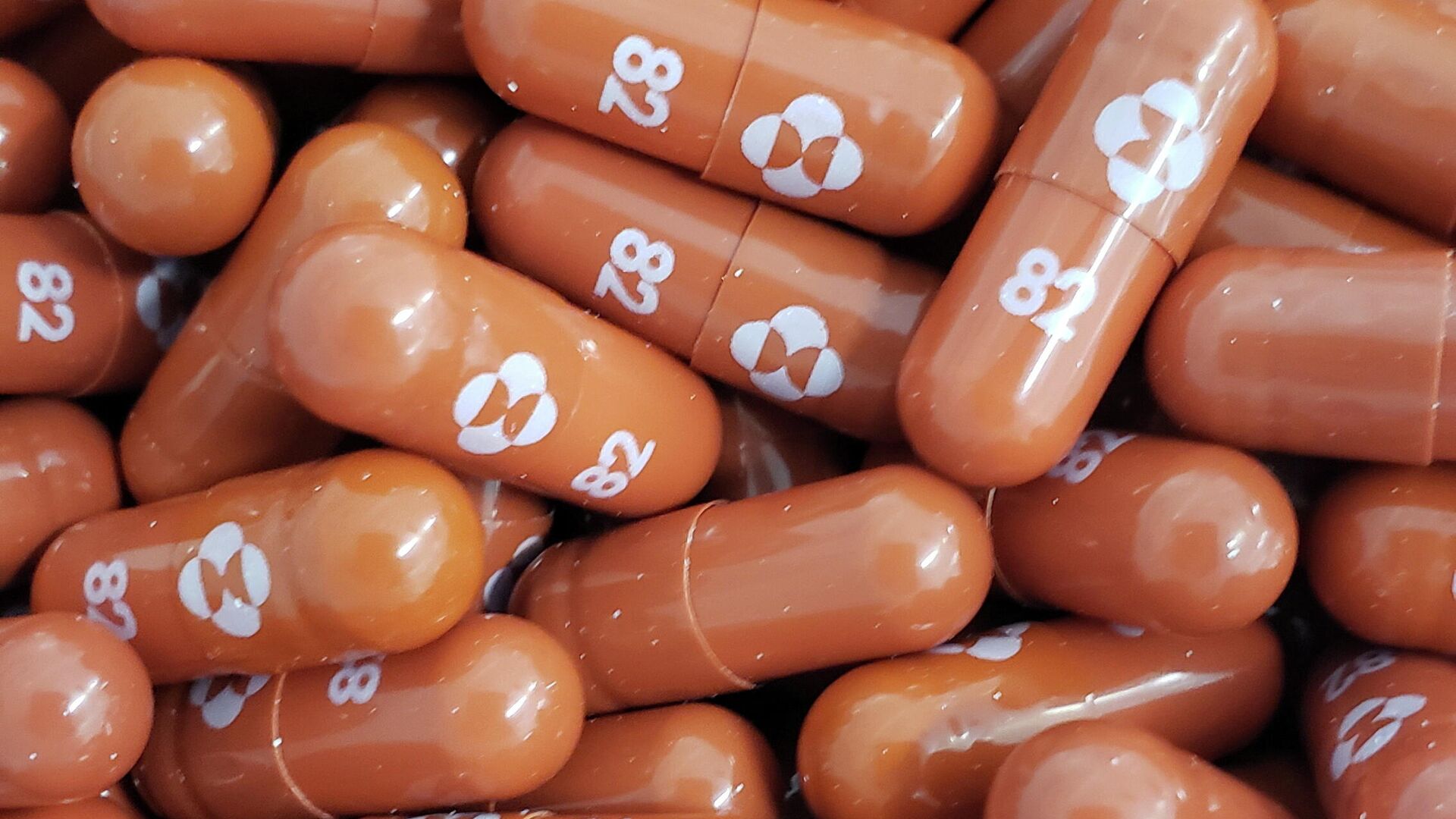
Federal health regulators have revealed that Molnupiravir, an investigational antiviral drug to treat COVID-19, is effective against the contagious disease, but the agency will seek input from experts regarding possible birth defects and other potential pregnancy issues associated with the pill. Scientists with the federal agency have identified several potential safety issues with the drug, including possible toxicity to developing fetuses and birth defects. This data was collected from trials using the pill in animals. The FDA's posting comes days before the agency's Antimicrobial Drugs Advisory Committee (AMDAC) is scheduled to meet to discuss Merck and Ridgeback's submission.The FDA will ask advisers during their November 30 meeting if the drug should be prescribed to all persons, or if exceptions should be made. The agency could decide to make the drug carry a warning about risks during pregnancy, but allow doctors to prescribe it in cases where the benefits outweigh the risks. Merck has said that the drug should not be used by children, according to the FDA, which agreed with the company. The pill, billed as a 5-day treatment, targets an enzyme that allows the coronavirus to make copies of itself and introduces errors to the virus' genetic code. The FDA also raised concerns about the fact that Molnupiravir was found to alter the virus' spike protein, which allows it to penetrate host cells and cause infection. This could theoretically result in the formation of new variants of COVID-19. Merck has observed a 30% reduction in hospitalizations and deaths with the antiviral pill, according to data from 1,433 patients. Merck previously touted data from 775 patients in October that showed the drug had an approximately 50% efficacy rate.
Sputnik International
feedback@sputniknews.com
+74956456601
MIA „Rossiya Segodnya“
2021
News
en_EN
Sputnik International
feedback@sputniknews.com
+74956456601
MIA „Rossiya Segodnya“
Sputnik International
feedback@sputniknews.com
+74956456601
MIA „Rossiya Segodnya“
pill, covid-19, health, children, patients, pandemic
pill, covid-19, health, children, patients, pandemic
FDA Deems Merck Pill Effective Against COVID-19 as Experts Plan to Review Safety
Last month, former Food and Drug administrator Scott Gottlieb said that he believes the COVID-19 "pandemic phase" will come to an end with the approval of antiviral pills and vaccines for children under 12 years old. An oral treatment developed by Merck & Co. and Ridgeback Biotherapeutics has been submitted to the FDA for emergency authorization.
Federal health regulators have revealed that Molnupiravir, an investigational antiviral drug to treat COVID-19, is effective against the contagious disease, but the agency will seek input from experts regarding possible birth defects and other potential pregnancy issues associated with the pill.
Scientists with the federal agency have identified several potential safety issues with the drug, including possible toxicity to developing fetuses and birth defects. This data was collected from trials using the pill in animals.
The FDA's posting comes days before the agency's Antimicrobial Drugs Advisory Committee (AMDAC) is scheduled to meet to discuss Merck and Ridgeback's submission.
The FDA will ask advisers during their November 30 meeting if the drug should be prescribed to all persons, or if exceptions should be made. The agency could decide to make the drug carry a warning about risks during pregnancy, but allow doctors to prescribe it in cases where the benefits outweigh the risks.
"While the clinical safety data base was small, there were no major safety concerns identified," FDA reviewers concluded, as reported by the Associated Press.
Merck has said that the drug should not be used by children, according to the FDA, which agreed with the company.
The pill, billed as a 5-day treatment, targets an enzyme that allows the coronavirus to make copies of itself and introduces errors to the virus' genetic code.
The FDA also raised concerns about the fact that Molnupiravir was found to alter the virus' spike protein, which allows it to penetrate host cells and cause infection. This could theoretically result in the formation of new variants of COVID-19.
Merck has observed a 30% reduction in hospitalizations and deaths with the antiviral pill, according to data from 1,433 patients. Merck previously touted data from 775 patients in October that showed the drug had an approximately 50% efficacy rate.