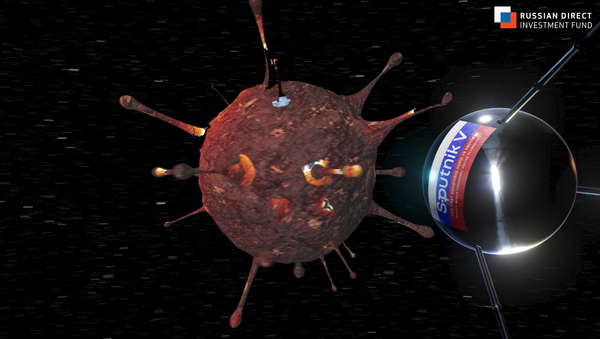
The Russian Direct Investment Fund, Russia’s sovereign wealth fund, has created a snazzy promotional clip for the Sputnik V COVID-19 vaccine.
The 38 second CGI video starts out with Earth being swallowed up by a giant coronavirus spore. Then, a Sputnik satellite reminiscent of Sputnik 1, the world’s first artificial Earth satellite launched in 1957 by the USSR, breaks through the virus and flies out into space, proceeding to circle the planet as the giant viral spore is gradually eradicated.
The clip is part of a press kit including photos, graphics and one other video showing the vaccine’s production which can be found on the vaccine’s official website.
The Sputnik V, formally named ‘Gam-COVID-Vak’, has already begun production. On Sunday, Gamaleya Institute director Alexander Ginzburg told Sputnik that mass vaccination using the Sputnik V vaccine should start in Russia in about a month’s time.
Russia aims to manufacture 500 million doses of the vaccine over the next year, with production and clinical trials expected to take place in several other countries as well.
The adenoviral vector vaccine consists of two shots, the second of which is injected three weeks after the first one.
Sputnik V was tested on 76 volunteers at two separate institutions – the Moscow-based Sechenov University and the Russian defence ministry’s Burdenko Main Military Clinical Hospital. Phase III trials involving tens of thousands of volunteers are to be carried out in the coming weeks. More information on the vaccine’s pre-clinical and clinical trials is expected to be published in the coming week to dispel observers’ fears regarding the vaccine’s safety.