https://sputnikglobe.com/20220216/pfizer-funding-fda-to-approve-annual-covid-jab-exec-tells-project-veritas-1093085203.html
Pfizer Funding FDA to Approve Annual COVID Jab, Exec Tells Project Veritas
Pfizer Funding FDA to Approve Annual COVID Jab, Exec Tells Project Veritas
Sputnik International
Big Pharma firm Pfizer and its German partner BioNtech are seeking emergency approval in the US for the use of their high-priced COVID-19 vaccine in toddlers... 16.02.2022, Sputnik International
2022-02-16T15:50+0000
2022-02-16T15:50+0000
2022-08-06T13:28+0000
project veritas
food and drug administration (fda)
joe biden
pfizer
big pharma
covid-19
coronavirus
us
https://cdn1.img.sputnikglobe.com/img/07e6/02/10/1093084205_0:121:3071:1848_1920x0_80_0_0_a9924f4af79462ff137e329d9ef181d4.jpg
A whistleblower has revealed drug firms that fund the US Food and Drug Administration (FDA) want it to approve annual COVID-19 jabs as a "recurring fountain of revenue".Christopher Cole, executive officer of the FDA's Medical Countermeasures initiative (MCMi), also told investigative journalism collective Project Veritas that US President Joe Biden backs the yet-to-be-announced plan for yearly boosters.The MCMi is tasked with preparing responses to terrorist attacks using biological, chemical, nuclear or radiological weapons, and for outbreaks of new infectious diseases like COVID-19.Cole said his office "clears all the emergency approvals" of vaccines, as was done for COVID-19 shots to get them into use before clinical trials, which normally take years, were complete. The executive exposed the glaring conflict of interest in how the FDA, the federal government agency responsible for approving medical treatments and food standards, is funded."The drug companies, the food companies, the vaccine companies, they pay us hundreds of millions of dollars a year to hire and keep the reviewers to approve their products."The whistleblower also said that from what he had heard, there was no way the FDA was "not going to approve" Pfizer coronavirus shots for children from six months up to five years. "It's going to be be a gradual thing. Schools are going to mandate it," Cole said."I don't completely agree with their... the process. They're looking to inoculate kids under five years old. Between six months and five years old."But he warned that not enough testing had been done to guarantee the safety of the Pfizer mRNA vaccine, which uses the body's cellular mechanisms to create copies of the virus' surface spike protein, in small children.The executive clarified that a lower bar was set for effectiveness and safety of emergency-approved vaccines.The FDA denied Cole's assertion he and his department "expedite approval" of vaccines in emergencies, while even attempting to question whether he was the man in the interview."The person purportedly in the video does not work on vaccine matters and does not represent the views of the FDA," the agency said in a statement.
https://sputnikglobe.com/20220202/pfizer-biontech-say-seeking-emergency-authorization-for-covid-19-vaccine-for-kids-under-5-1092670756.html
Sputnik International
feedback@sputniknews.com
+74956456601
MIA „Rossiya Segodnya“
2022
James Tweedie
https://cdn1.img.sputnikglobe.com/img/07e4/08/1c/1080307270_0:3:397:400_100x100_80_0_0_7777393b9b18802f2e3c5eaa9cbcc612.png
James Tweedie
https://cdn1.img.sputnikglobe.com/img/07e4/08/1c/1080307270_0:3:397:400_100x100_80_0_0_7777393b9b18802f2e3c5eaa9cbcc612.png
News
en_EN
Sputnik International
feedback@sputniknews.com
+74956456601
MIA „Rossiya Segodnya“
Sputnik International
feedback@sputniknews.com
+74956456601
MIA „Rossiya Segodnya“
James Tweedie
https://cdn1.img.sputnikglobe.com/img/07e4/08/1c/1080307270_0:3:397:400_100x100_80_0_0_7777393b9b18802f2e3c5eaa9cbcc612.png
project veritas, food and drug administration (fda), joe biden, pfizer, big pharma, covid-19, coronavirus, us
project veritas, food and drug administration (fda), joe biden, pfizer, big pharma, covid-19, coronavirus, us
Pfizer Funding FDA to Approve Annual COVID Jab, Exec Tells Project Veritas
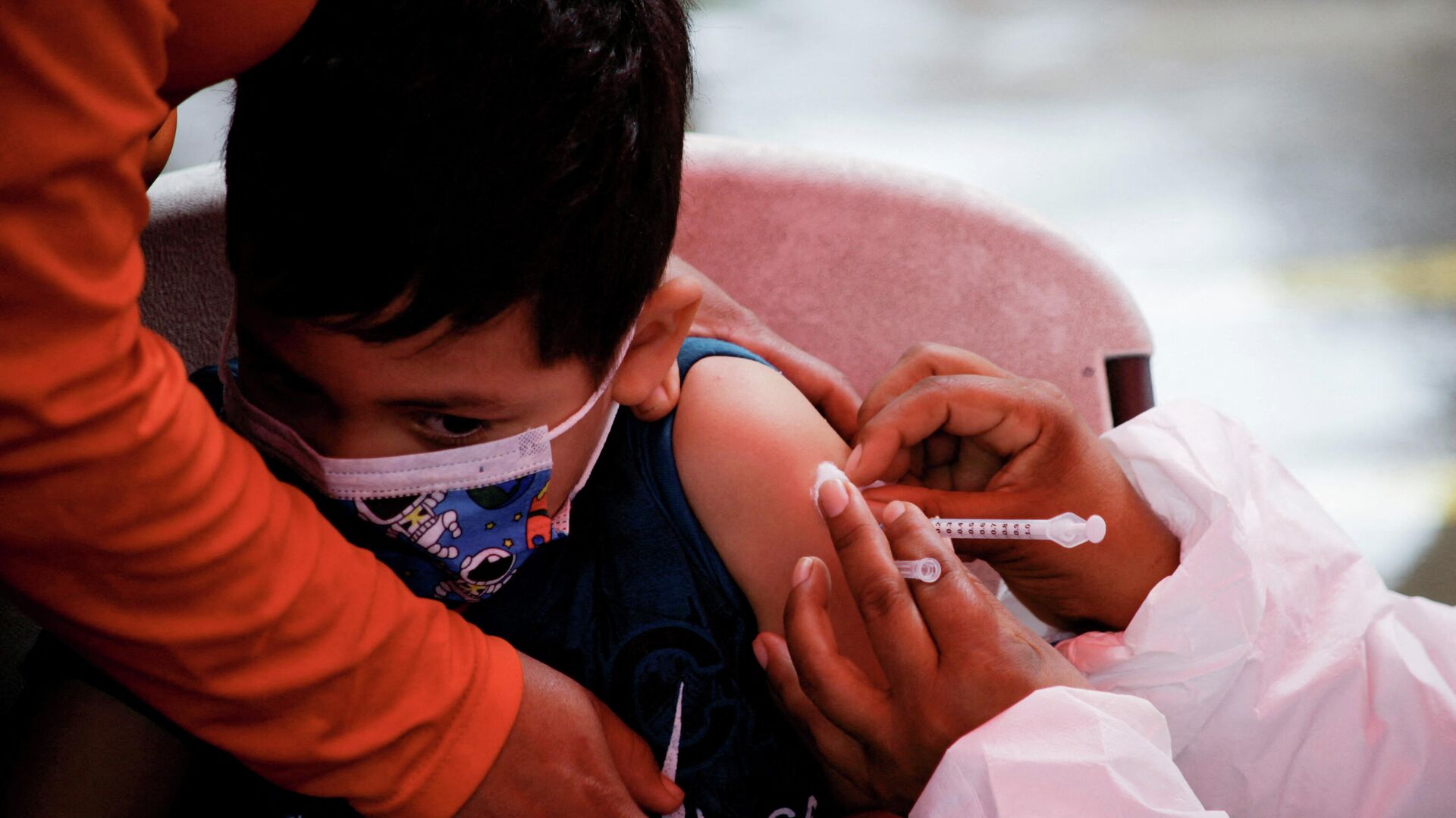
15:50 GMT 16.02.2022 (Updated: 13:28 GMT 06.08.2022) Big Pharma firm Pfizer and its German partner BioNtech are seeking emergency approval in the US for the use of their high-priced COVID-19 vaccine in toddlers and babies as young as six months old.
A whistleblower has revealed drug firms that fund the US Food and Drug Administration (FDA) want it to approve annual COVID-19 jabs as a "recurring fountain of revenue".
Christopher Cole, executive officer of the FDA's Medical Countermeasures initiative (MCMi), also told investigative journalism collective
Project Veritas that US President Joe Biden backs the yet-to-be-announced plan for yearly boosters.
"Biden wants to inoculate as many people as possible," Cole told an undercover reporter in hidden camera footage. "So you'll have to get an annual shot. It hasn’t been formally announced yet ‘cause they don’t want to, like, rile everyone up."
The
MCMi is tasked with preparing responses to terrorist attacks using biological, chemical, nuclear or radiological weapons, and for outbreaks of new infectious diseases like COVID-19.
Cole said his office "clears all the emergency approvals" of vaccines, as was done for COVID-19 shots to get them into use before clinical trials, which normally take years, were complete.
The executive exposed the glaring conflict of interest in how the FDA, the federal government agency responsible for approving medical treatments and food standards, is funded.
"The drug companies, the food companies, the vaccine companies, they pay us hundreds of millions of dollars a year to hire and keep the reviewers to approve their products."
"There’s a money incentive for Pfizer and the drug companies to promote additional vaccinations," Cole added. “It’ll be recurring fountain of revenue. It might not be that much initially, but it’ll recurring if they can get every person required at an annual vaccine, that is a recurring return of money going into their company."
The whistleblower also said that from what he had heard, there was no way the FDA was "not going to approve" Pfizer coronavirus shots for children from six months up to five years. "It's going to be be a gradual thing. Schools are going to mandate it," Cole said.
"I don't completely agree with their... the process. They're looking to inoculate kids under five years old. Between six months and five years old."
But he warned that not enough testing had been done to guarantee the safety of the Pfizer mRNA vaccine, which uses the body's cellular mechanisms to create copies of the virus' surface spike protein, in small children.
"All the tests aren't there... you can't provide the assurity [sic] for parents," Cole stressed. "There's always a chance of a long-term effect, especially with someone younger."
2 February 2022, 00:52 GMT
The executive clarified that a lower bar was set for
effectiveness and safety of emergency-approved vaccines.
"The efficacy data doesn't have to be as high, Cole said. "The standard is on emergency use authorisations is that it does more benefit than harm."
The FDA denied Cole's assertion he and his department "expedite approval" of vaccines in emergencies, while even attempting to question whether he was the man in the interview.
"The person purportedly in the video does not work on vaccine matters and does not represent the views of the FDA," the agency said in a statement.